
Police report details grisly crime scene in Greenfield
SUNDERLAND — Police located a cabin in the woods around Clark Mountain Road Thursday afternoon that is believed to belong to Taaniel Herberger-Brown, the suspect accused of murdering a man at 92 Chapman St. in Greenfield and storing his body in a...

Frontier Regional School students appeal to lower voting age
When Sunderland residents head to their Annual Town Meeting on Friday, they’ll vote on the town’s budget, some bylaw changes and a citizen’s petition with a unique twist, as the residents who brought the article forward can’t actually vote on...
Most Read
 Police report details grisly crime scene in Greenfield
Police report details grisly crime scene in Greenfield
 On The Ridge with Joe Judd: What time should you turkey hunt?
On The Ridge with Joe Judd: What time should you turkey hunt?
 New buyer of Bernardston’s Windmill Motel looks to resell it, attorney says
New buyer of Bernardston’s Windmill Motel looks to resell it, attorney says
 Greenfield man arrested in New York on murder charge
Greenfield man arrested in New York on murder charge
 Man allegedly steals $100K worth of items from Northampton, South Deerfield businesses
Man allegedly steals $100K worth of items from Northampton, South Deerfield businesses
 Joannah Whitney of Greenfield wins 33rd annual Poet’s Seat Poetry Contest
Joannah Whitney of Greenfield wins 33rd annual Poet’s Seat Poetry Contest
Editors Picks
 Montague Notebook: April 24, 2024
Montague Notebook: April 24, 2024
 Monroe election sees no contests, some positions without candidates
Monroe election sees no contests, some positions without candidates
 Business Briefs: April 26, 2024
Business Briefs: April 26, 2024
 PHOTO: Spectacular spire
PHOTO: Spectacular spire
Sports

Track & field: Greenfield, Mohawk Trail split dual meet
It was a dual meet split for the Greenfield and Mohawk Trail track and field teams on Thursday.The Greenfield boys captured a 90-54 victory over the Warriors while the Mohawk Trail girls snagged a 123-18 victory over the Wave.Here are the winners from...
 Country Club of Greenfield holding Youth Golf Camp June 24-27
Country Club of Greenfield holding Youth Golf Camp June 24-27
Opinion

My Turn: A moral justification for civil disobedience to abortion bans
Over the last several years, in response to abortion bans and restrictions, advocates around the country have developed an alternative supply network for abortion pills outside of the medical system and the law.As a lawyer and law-abiding citizen, I...
 Guest columnist Rudy Perkins: Dangerous resolution on Iran
Guest columnist Rudy Perkins: Dangerous resolution on Iran
 Guest columnists Ellen Attaliades and Lynn Ireland: Housing crisis is fueling the human services crisis
Guest columnists Ellen Attaliades and Lynn Ireland: Housing crisis is fueling the human services crisis
 My Turn: April is second chance month
My Turn: April is second chance month

Business

Goddard finds ‘best location’ in Shelburne Falls with new Watermark Gallery space
SHELBURNE FALLS — The 20 Bridge St. space that formerly housed Molly Cantor’s pottery now displays Watermark Gallery’s eclectic collection of modern art pieces.Local artist Laurie Goddard opened Watermark Gallery last month as her seventh location in...
 New buyer of Bernardston’s Windmill Motel looks to resell it, attorney says
New buyer of Bernardston’s Windmill Motel looks to resell it, attorney says
 New owners look to build on Thomas Memorial Golf & Country Club’s strengths
New owners look to build on Thomas Memorial Golf & Country Club’s strengths
 Cleary Jewelers plans to retain shop at former Wilson’s building until 2029
Cleary Jewelers plans to retain shop at former Wilson’s building until 2029
Arts & Life

Sounds Local: A rock circus returns to Turners Falls: The Slambovian Circus of Dreams brings the fun Friday night at the Shea
The circus is coming to town, and not the kind with elephants and clowns. I’m talking about the Slambovian Circus of Dreams, a rock group from the Hudson Valley in New York that makes music as unique as its name. The band, which has frequently...
Obituaries
 Dorothy Masterson Bennett
Dorothy Masterson Bennett
Conway, MA - Dorothy Masterson, the daughter of John Cochran Masterson and Dorothy Goodwin Masterson, was born January 15, 1932 in the Mary Alley Hospital on Franklin Street, Marblehead. She died April 20, 2024, age 92 years, in her bed... remainder of obit for Dorothy Masterson Bennett
 Audrey McKemmie
Audrey McKemmie
Greenfield, MA - Audrey A. McKemmie of Greenfield and formerly of Amherst passed away on April 23, 2024, after a period of declining health. Born in Greenfield, December 12, 1930, she was the only child of John and Susie (Hayden) Kolink... remainder of obit for Audrey McKemmie
 Reita G. Wheeler
Reita G. Wheeler
Colrain, MA - Reita G. (Sell) Wheeler, 77, of East Colrain Rd., passed away on Wednesday, April 24, 2024 at Baystate Franklin Medical Center in Greenfield. Reita was born in Westfield, MA on November 25, 1946 the daughter of Gustave... remainder of obit for Reita G. Wheeler
 Constance Julia Dargis
Constance Julia Dargis
Constance Julia (LaMountain) Dargis Montague Center, MA - Constance "Connie" Julia Dargis, 98, formerly of Turners Falls MA, suffered an unexpected illness on November 13th and passed away peacefully on November 16, 2023 at the home of her ... remainder of obit for Constance Julia Dargis

 Montague Cemetery Commission dedicating town’s first green burial site
Montague Cemetery Commission dedicating town’s first green burial site
 Ethics Commission raps former Leyden police chief, captain for conflict of interest violations
Ethics Commission raps former Leyden police chief, captain for conflict of interest violations
 Federal probe targets UMass response to anti-Arab incidents
Federal probe targets UMass response to anti-Arab incidents
 New Salem event will turn gun parts into gardening tools, jewelry
New Salem event will turn gun parts into gardening tools, jewelry
 Advancing water treatment: UMass startup Elateq Inc. wins state grant to deploy new technology
Advancing water treatment: UMass startup Elateq Inc. wins state grant to deploy new technology
 Sunderland Public Library hosting 20th birthday party for its building
Sunderland Public Library hosting 20th birthday party for its building
 Real Estate Transactions: April 26, 2024
Real Estate Transactions: April 26, 2024
 Shelburne fair to explore energy-efficient options for homeowners
Shelburne fair to explore energy-efficient options for homeowners
 Erving rejects trade school’s incomplete proposal for mill reuse
Erving rejects trade school’s incomplete proposal for mill reuse
 High schools: Amy Mihailicenco, Greenfield girls tennis edge Turners Falls (PHOTOS)
High schools: Amy Mihailicenco, Greenfield girls tennis edge Turners Falls (PHOTOS) Softball: Turners edges Tech, Greenfield’s Paulin notches 100th hit, Mahar win first game since 2019
Softball: Turners edges Tech, Greenfield’s Paulin notches 100th hit, Mahar win first game since 2019 High schools: Lilly Ross records 100th career hit in Franklin Tech’s win over Northampton
High schools: Lilly Ross records 100th career hit in Franklin Tech’s win over Northampton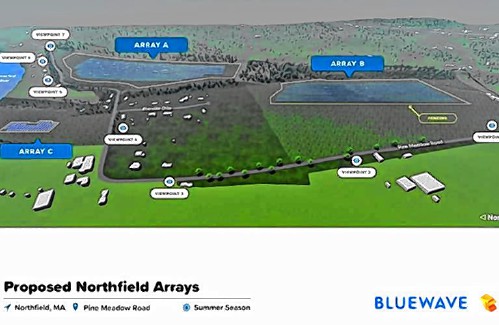 My Turn: Industrial solar installations are overwhelming Northfield neighborhood
My Turn: Industrial solar installations are overwhelming Northfield neighborhood New Realtor Association CEO looks to work collaboratively to maximize housing options
New Realtor Association CEO looks to work collaboratively to maximize housing options Rescuing food and feeding people: Rachel’s Table programs continue to expand throughout western Mass
Rescuing food and feeding people: Rachel’s Table programs continue to expand throughout western Mass A day to commune with nature: Western Mass Herbal Symposium will be held May 11 in Montague
A day to commune with nature: Western Mass Herbal Symposium will be held May 11 in Montague Speaking of Nature: ‘Those sound like chickens’: Wood frogs and spring peepers are back — and loud as ever
Speaking of Nature: ‘Those sound like chickens’: Wood frogs and spring peepers are back — and loud as ever Hitting the ceramic circuit: Asparagus Valley Pottery Trail turns 20 years old, April 27-28
Hitting the ceramic circuit: Asparagus Valley Pottery Trail turns 20 years old, April 27-28
