
Joannah Whitney of Greenfield wins 33rd annual Poet’s Seat Poetry Contest
GREENFIELD — After a few years submitting her work to the annual Poet’s Seat Poetry Contest, Greenfield poet and freelance writer Joannah Whitney placed first among the contest’s adult finalists Tuesday night at the Greenfield Public Library. The...

New buyer of Bernardston’s Windmill Motel looks to resell it, attorney says
BERNARDSTON — The Windmill Motel was sold at auction earlier this month to a private out-of-state lender.The motel at 497 Northfield Road, which was opened by Kolman Nemes in 1981, was bought for $750,000 on April 10, according to Marianne Sullivan,...
Most Read
 Greenfield man arrested in New York on murder charge
Greenfield man arrested in New York on murder charge
 Man allegedly steals $100K worth of items from Northampton, South Deerfield businesses
Man allegedly steals $100K worth of items from Northampton, South Deerfield businesses
 Greenfield Police Logs: April 9 to April 17, 2024
Greenfield Police Logs: April 9 to April 17, 2024
 Former Leyden police chief Daniel Galvis charged with larceny
Former Leyden police chief Daniel Galvis charged with larceny
 Shea Theater mural artist chosen out of 354 applicants
Shea Theater mural artist chosen out of 354 applicants
 Millers Meadow idea would ‘completely transform’ Colrain Street lot in Greenfield
Millers Meadow idea would ‘completely transform’ Colrain Street lot in Greenfield
Editors Picks
 Sounds Local: A rock circus returns to Turners Falls: The Slambovian Circus of Dreams brings the fun Friday night at the Shea
Sounds Local: A rock circus returns to Turners Falls: The Slambovian Circus of Dreams brings the fun Friday night at the Shea
 Monroe election sees no contests, some positions without candidates
Monroe election sees no contests, some positions without candidates
 Business Briefs: April 26, 2024
Business Briefs: April 26, 2024
 PHOTO: A valuable simulation
PHOTO: A valuable simulation
Sports

Country Club of Greenfield holding Youth Golf Camp June 24-27
The Country Club of Greenfield is holding its Youth Golf Camp from June 24-27. The camp will be capped at 25 children on a first come, first serve basis. The fee is $170 for the first family member and $140 for each additional family member. The camp...
 High schools: Lilly Ross records 100th career hit in Franklin Tech’s win over Northampton
High schools: Lilly Ross records 100th career hit in Franklin Tech’s win over Northampton
 Baseball: Wyatt Edes’ walk-off single lifts Frontier past Amherst 6-5
Baseball: Wyatt Edes’ walk-off single lifts Frontier past Amherst 6-5
 On The Ridge with Joe Judd: What time should you turkey hunt?
On The Ridge with Joe Judd: What time should you turkey hunt?
 High Schools: Jakhia Williams propels Turners girls track past Athol
High Schools: Jakhia Williams propels Turners girls track past Athol
Opinion

My Turn: April is second chance month
April is Second Chance Month, a time to focus on how our community can support our neighbors in central and western Massachusetts returning from incarceration.As attorneys with Community Legal Aid’s CORI/Re-Entry Unit, we see the life-changing power...
 My Turn: Judgmental about malaise
My Turn: Judgmental about malaise
 Ahmad Esfahani: Aiding and abetting and middle school angst
Ahmad Esfahani: Aiding and abetting and middle school angst
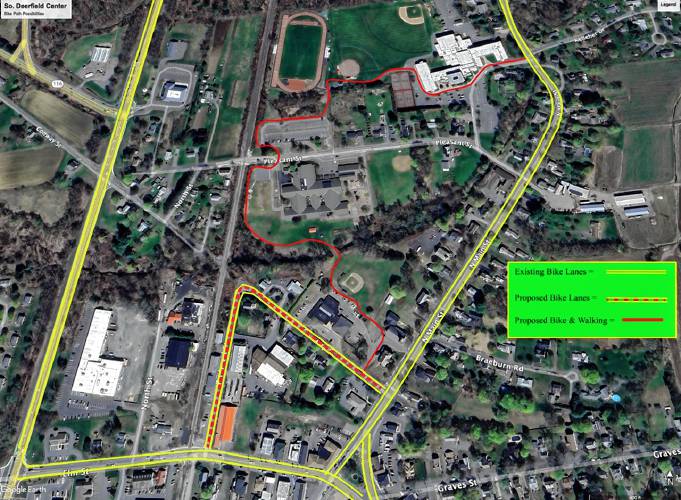 Greg Franceschi: Support bike lanes and walking paths in South Deerfield
Greg Franceschi: Support bike lanes and walking paths in South Deerfield
 Laurie Benoit: Volunteers needed for cleanup of Buckland cemeteries
Laurie Benoit: Volunteers needed for cleanup of Buckland cemeteries

Business

New Realtor Association CEO looks to work collaboratively to maximize housing options
SPRINGFIELD — As the Realtor Association of Pioneer Valley’s new CEO arrives to a Massachusetts housing market plagued by high prices and a lack of stock, he aims to work alongside elected officials to maximize the availability of different kinds of...
 New owners look to build on Thomas Memorial Golf & Country Club’s strengths
New owners look to build on Thomas Memorial Golf & Country Club’s strengths
 Cleary Jewelers plans to retain shop at former Wilson’s building until 2029
Cleary Jewelers plans to retain shop at former Wilson’s building until 2029
 Tea Guys of Whately owes $2M for breach of contract, judge rules
Tea Guys of Whately owes $2M for breach of contract, judge rules
 Primo Restaurant & Pizzeria in South Deerfield under new ownership
Primo Restaurant & Pizzeria in South Deerfield under new ownership
Arts & Life

Rescuing food and feeding people: Rachel’s Table programs continue to expand throughout western Mass
My great-grandmother’s oak dining table has graced my kitchen ever since my parents built the house in the 1980s. Looking at it makes me happy. The table — actually, almost any table — signifies history, nourishment, family and community.The...
Obituaries
 Constance Julia Dargis
Constance Julia Dargis
Constance Julia (LaMountain) Dargis Montague Center, MA - Constance "Connie" Julia Dargis, 98, formerly of Turners Falls MA, suffered an unexpected illness on November 13th and passed away peacefully on November 16, 2023 at the home of her ... remainder of obit for Constance Julia Dargis
 John H. Stevens Jr.
John H. Stevens Jr.
John H. Stevens, Jr. Gill, MA - John H. Stevens, Jr., 69, of Main Road died Monday 4/22/24 at home. He was born in Greenfield on December 9, 1954, one of nine children of John H. and Evelyn (Bassett) Stevens. John worked as a papermaker... remainder of obit for John H. Stevens Jr.
 Maryjean DelRosso
Maryjean DelRosso
Heath, MA - Maryjean (Sullivan) DelRosso passed away peacefully on April 19, 2024 at Baystate Medical Center in Springfield after a long illness. She is survived by her devoted husband, Joseph DelRosso. Maryjean is also survived by thr... remainder of obit for Maryjean DelRosso
 Carol Butler Watelet
Carol Butler Watelet
Greenfield, MA - Carol (Virginia) Soucie Butler Watelet, 85, passed away suddenly and peacefully on April 20, 2024, joining the love of her life, Mr. Wonderful, Robert Watelet who passed away 2 years earlier. Carol was born on March 17,... remainder of obit for Carol Butler Watelet

 New Salem event will turn gun parts into gardening tools, jewelry
New Salem event will turn gun parts into gardening tools, jewelry
 Advancing water treatment: UMass startup Elateq Inc. wins state grant to deploy new technology
Advancing water treatment: UMass startup Elateq Inc. wins state grant to deploy new technology
 Sunderland Public Library hosting 20th birthday party for its building
Sunderland Public Library hosting 20th birthday party for its building
 Real Estate Transactions: April 26, 2024
Real Estate Transactions: April 26, 2024
 Shelburne fair to explore energy-efficient options for homeowners
Shelburne fair to explore energy-efficient options for homeowners
 Erving rejects trade school’s incomplete proposal for mill reuse
Erving rejects trade school’s incomplete proposal for mill reuse
 Parents question handling of threat at Erving Elementary School
Parents question handling of threat at Erving Elementary School
 Guest columnists Ellen Attaliades and Lynn Ireland: Housing crisis is fueling the human services crisis
Guest columnists Ellen Attaliades and Lynn Ireland: Housing crisis is fueling the human services crisis
 William Strickland, a longtime civil rights activist, scholar and friend of Malcolm X, has died
William Strickland, a longtime civil rights activist, scholar and friend of Malcolm X, has died
 A day to commune with nature: Western Mass Herbal Symposium will be held May 11 in Montague
A day to commune with nature: Western Mass Herbal Symposium will be held May 11 in Montague Speaking of Nature: ‘Those sound like chickens’: Wood frogs and spring peepers are back — and loud as ever
Speaking of Nature: ‘Those sound like chickens’: Wood frogs and spring peepers are back — and loud as ever Hitting the ceramic circuit: Asparagus Valley Pottery Trail turns 20 years old, April 27-28
Hitting the ceramic circuit: Asparagus Valley Pottery Trail turns 20 years old, April 27-28 Best Bites: A familiar feast: The Passover Seder traditions and tastes my family holds dear
Best Bites: A familiar feast: The Passover Seder traditions and tastes my family holds dear
